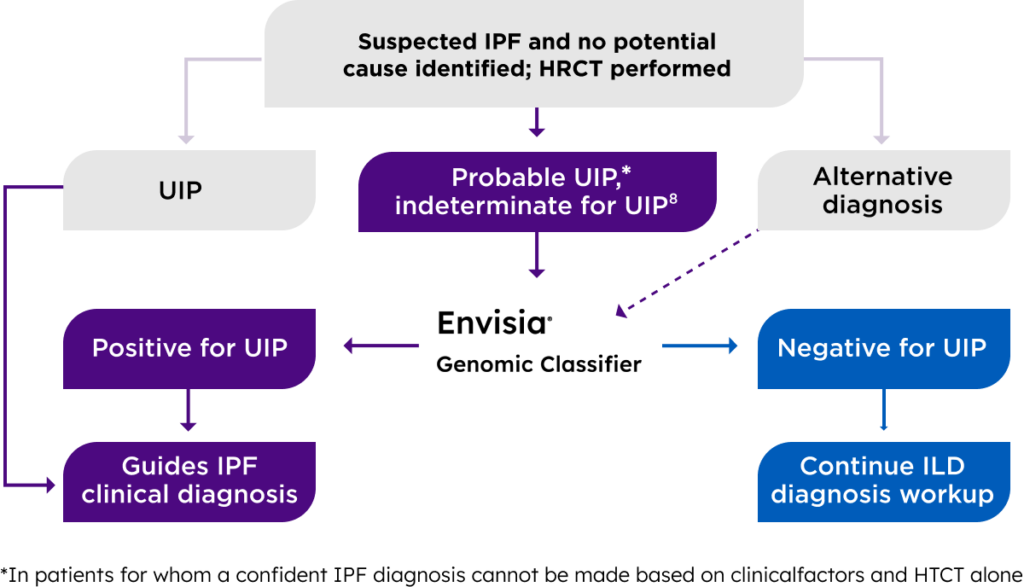
Envisia® detects a genomic UIP pattern to improve ILD diagnostic and prognostic confidence
ABOUT ENVISIA®
What is the test?
Why is usual interstitial pneumonia (UIP) important for suspected interstitial lung diseases (ILD)?
UIP is a critical factor in differentiating idiopathic pulmonary fibrosis (IPF) from other ILDs to determine appropriateness of antifibrotic treatment. Additionally, UIP is associated with poor prognosis across ILD subtypes.
What is Envisia?
Envisia classifier detects the genomic pattern of UIP using transbronchial biopsy (TBB) samples to help guide diagnostic and prognostic decisions. Envisia results must be interpreted in the context of other clinical factors such as demographics, clinical history, and high resolution CT (HRCT) findings.
Watch brief overview of the Envisia classifier
Learn how to improve ILD diagnostic confidence with the Envisia classifier.
Who is a good candidate for the Envisia test?
Clinical Factors:
Have no known causes with suspected ILD
HRCT:
Probable UIP or indeterminate for UIP
Tolerability:
Able to tolerate standard TBBx procedure
Envisia classifier results indicate either a positive or negative for UIP
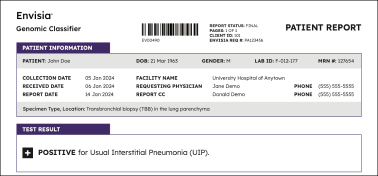
Positive results
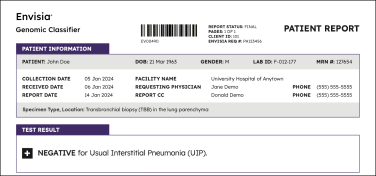
Negative results
Sample collection and ordering
Samples are collected through standard forceps transbronchial biopsy (TBB). Test results delivered within two weeks to your computer or phone.
References
1. Raghu G, et al. Lancet Respiratory Medicine. Apr 2019.

